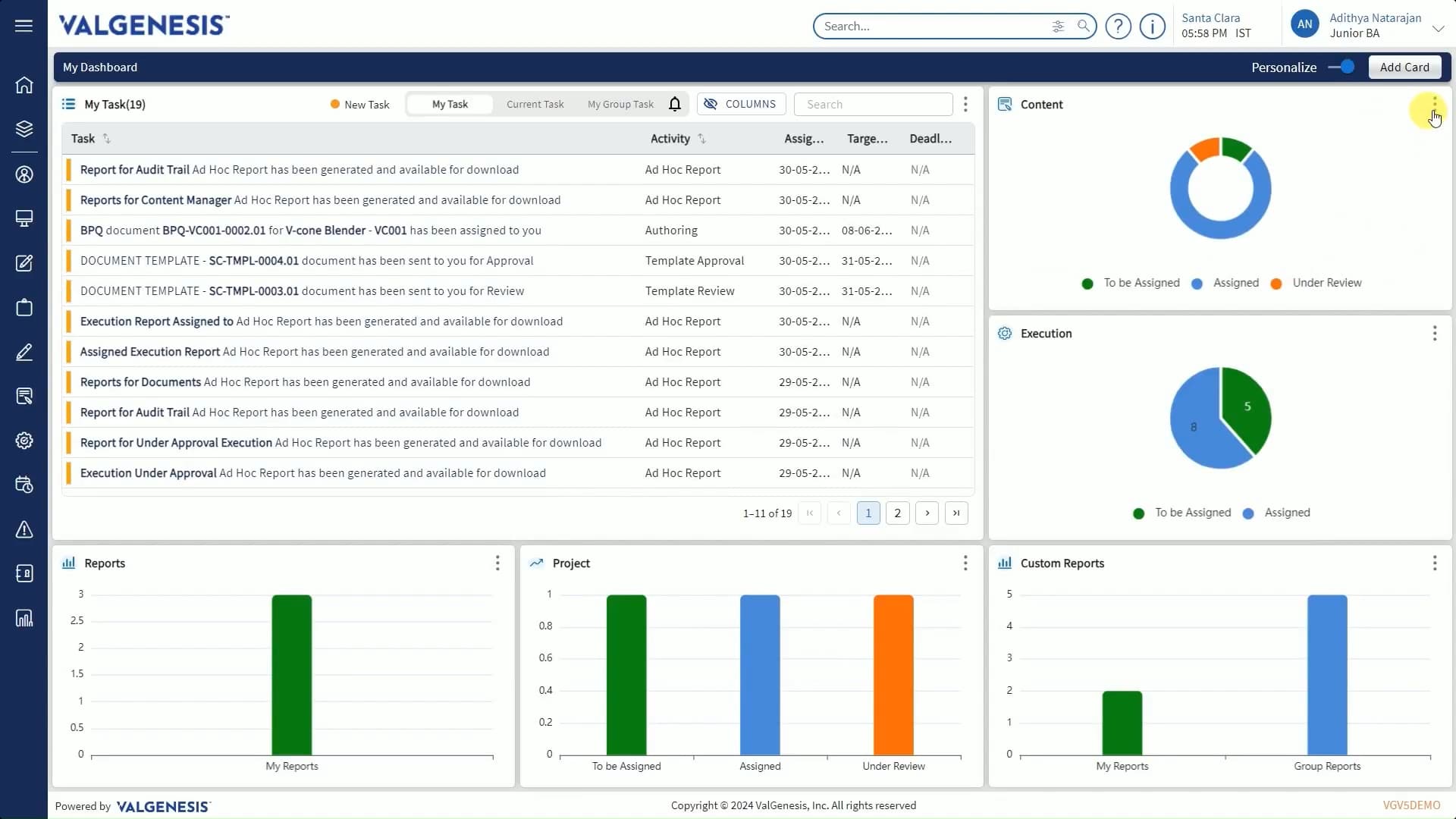
ValGenesis VLMS
The leader in digitizing validation
Setting the digital validation standard for 20 years, ValGenesis VLMS is used by 30 of the top 50 global life sciences companies.
ValGenesis VLMS
The gold standard of standardization
Strengthen your compliance stance and lower the cost of quality with enforced standardization and absolute data integrity.
ValGenesis VLMS
Knowledge integrity to data integrity
Gain total peace of mind with a single source of validation truth with documentation aligned to ALCOA+ standards.
ValGenesis VLMS
One platform, boundless scale
Start small or start global. VLMS scales effortlessly to support new systems, new sites, new products, new languages, new validation processes.
Digitize.
Standardize.
Harmonize.
ValGenesis VLMS is the Industry Standard for Digital Validation Across Life Sciences
Peerless in capability, VLMS provides a unified, data-centric platform for digitizing the entire validation lifecycle, enforcing standardization, ensuring data integrity, reducing risk, lowering the cost of quality, strengthening compliance, and more.
Gain 100% digital, risk-based validation in CSV/CSA, commissioning and qualification, facilities and equipment, and beyond.
Announcing VLMS 5.0
The newest release features a holistic reimagining of the user experience, pairing redesigned user flows, speed enhancements, a modern UI, and AI-powered productivity.
Digitize and Streamline All Validation Activities
for Complete Validation Lifecycle Management
C&Q for Equipment and Facilities
VLMS streamlines commissioning and qualification via digitized data collection, testing, and documentation, ensuring efficiency from installation to operational readiness.
CSV and CSA
VLMS digitizes and simplifies CSV and CSA processes with a risk-based approach that facilitates all testing methodologies and reduces documentation burden.
Cleaning Validation
VLMS automates residue testing, documentation, and reporting, with full support for impact assessments, continuous monitoring, and periodic reviews.
Analytical Method Validation
VLMS facilitates the assessment of parameters like accuracy, precision, and robustness, ensuring analytical methods are reliable across different laboratories and conditions.
Logbook Management
VLMS digitizes and standardizes logs for equipment usage, maintenance, calibration, and other GxP records, providing real-time access to data with offline data capture.
Process Validation
VLMS supports automatic data collection, analysis, and reporting throughout the lifecycle to manufacturing processes consistently produce products that meet quality standards.
First Look: VLMS 5.0
Get a first look at ValGenesis VLMS 5.0, the latest milestone release of the industry's leading digital validation solution.
Digital Validation
Sophia Vraka
Senior Solutions Engineer
ValGenesis VLMS Drives Leaps in Productivity
Faster
Authoring, executing, and approving validation protocols
Faster
Creating and maintaining traceability matrices
Faster
Performing, reviewing, and approving risk assessments
Faster
Tracking validation status
Faster
Preparing for audits and inspections
ValGenesis VLMS has tremendously reduced our manual work. It's contemporaneous, easy to execute, and we can quickly access executed documents. More importantly, it has improved compliance.
Rajesh Thempadiyill
Head of Quality for Digital Transformation & Compliance
Dr Reddy's Laboratories
Industry Leaders Lead with ValGenesis VLMS

Talk to us to see how ValGenesis can support your digital transformation.